Electron geometry chart and examples3/10/2024 Bonds drawn with a solid triangle, or wedge, represent bonded atoms coming out of the page/screen towards the observer bonds drawn with dashed triangle, or dash, represent bonded atoms going into the page/screen away from the observer. Bonds drawn as simple lines are located in the plane of the page or screen. Chemists use dash and wedge notation to draw 3D molecules. Representing three dimensions on 2D paper (or computer screen) is challenging. The Lewis structure is misleading because it depicts methane as a planar ‘plus-shaped’ molecule, yet we know that methane is a non-planar, tetrahedral molecule. Methane (CH 4) depicted by a Lewis structure (left) and dash and wedge notation (right). Depicted on the left in Figure 1 is the Lewis structure for methane (CH 4) showing the central carbon atom singly bonded to four hydrogen atoms: Figure 1. 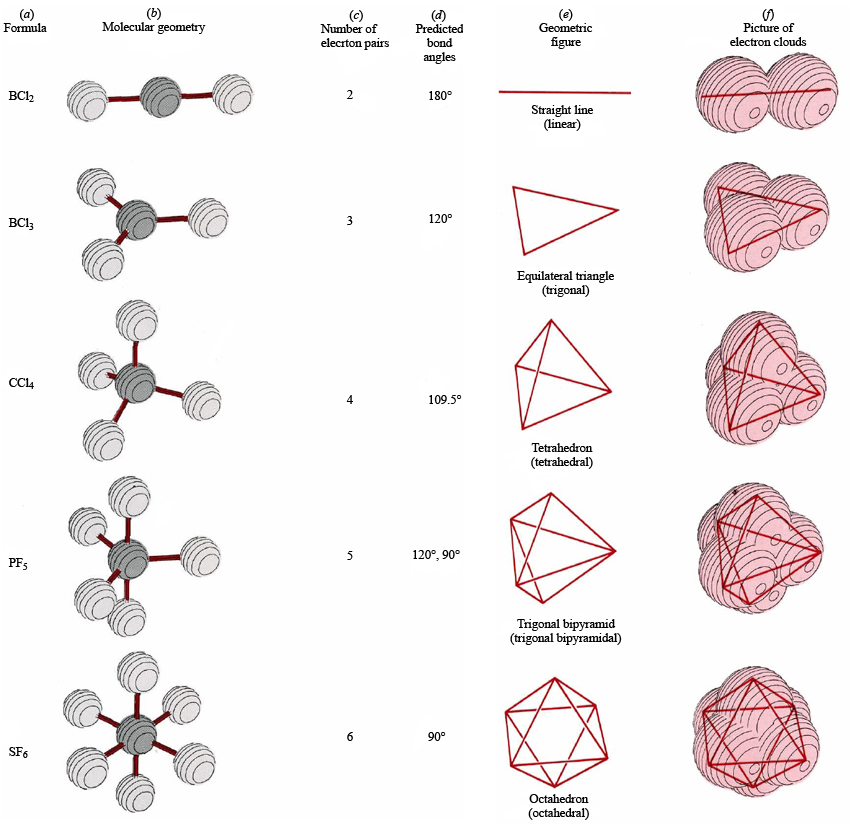
The Lewis structure gives us meaningful information about the bonds between atoms, but Lewis structures do not depict how the molecule exists in three-dimensions. | Key Concepts and Summary | Glossary | End of Section Exercises | Lewis Structures with Wedge-Dash Notation | Lewis Structures with Wedge-Dash Notations | Draw and interpret 3-dimensional representations of molecules using “dashed” and “wedge” bonds and estimate bond angles.Molecular Geometry | Predicting Electron-pair Geometry and Molecular Geometry | VSEPR Review Chart | | VSEPR Theory | Electron-pair Geometry vs. Predict molecular shape as determined by Valence Shell Electron Pair Repulsion Theory (VSEPR).
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
